Meat Science Review
Effect of direct-fed microbial on Enterococci antibiotic resistance
Studying the antibiotic resistance of Enterococcus sp. in cattle feces as impacted by dietary supplementation with Lactobacillus salivarius L28.

This study was designed to determine the antibiotic resistance patterns of Enterococcus sp. isolated from feedlot cattle supplemented with a novel direct-fed microbial (DFM) Lactobacillus salivarius (L28) in replacement of tylosin in finishing diets of feedlot steers.
Antibiotics have been used widely in veterinary and human health for more than 50 years (Dibner and Richards, 2005). The broad use of antibiotics in animal agriculture, specifically for growth promotion, has increased concerns that it could contribute to the increased prevalence of antimicrobial-resistant (AMR) bacteria. Using antibiotics over time can potentially result in significant pools of resistance genes among bacteria, including foodborne pathogens (Noyes et al., 2016). Therefore, further investigation of appropriate alternatives to the sub-therapeutic dosages of antibiotics in feedlot operations is necessary. Alternatives such as direct-fed microbials may be an effective solution. The use of DFM for ruminants has shown to enhance milk production in dairy cows, and for beef cattle, improve feed efficiency and increase average daily gain (Krehbiel et al., 2010). Research from Peterson et al. (2006) has also shown a reduction in fecal shedding of Escherichia coli O157:H7 in feedlot cattle with supplementation of a DFM.
Enterococcus sp. may not be a foodborne pathogen of high concern, but it is ubiquitous in nature and commonly found in the digestive tract of humans and animals. Enterococci are of medical importance and a leading cause of health care-associated infections in humans (Sood et al., 2008). Furthermore, they are important organisms because of their capability to develop antimicrobial-resistant genes and transfer those genes to other gram-positive bacteria that are potentially of a greater foodborne illness concern (Jackson et al., 2011).
A newly isolated direct-fed microbial, L. salivarius L28 was used in this study. Three feeding treatments based on conventional high-concentrate diets were fed to cattle for finishing prior to harvest: base diet (no DFM, tylosin or monensin), MonPro (DFM with monensin but no tylosin) and a control (tylosin and monensin). A total of 36 composite fecal samples, from three animals per pen, were collected after 56 days of feeding. Isolates of Enterococcus were obtained from fecal samples through conventional culturing techniques. Three isolates from each composite fecal sample were tested for antibiotic resistance. Data analysis was done by using a Chi-Squared test to compare resistance patterns across treatment group with a significant value of 5 percent.
Results from this study showed that Enterococcus sp. was isolated from 100 percent of fecal samples collected. After 56 days of feeding, all of the control groups’ isolates were resistant to at least one antibiotic, and 66 percent were multi-drug resistant (MDR) to three or more antibiotics. Isolates from the base treatment group on day 56 exhibited 97 percent resistance to one drug and 28 percent were MDR. Ninety-four percent of isolates in the MonPro group showed resistance on day 56 and 47 percent of isolates were MDR. The percent of isolates with MDR differed significantly between treatments (p = 0.004). Specific antibiotics of concern are listed in Table 1.
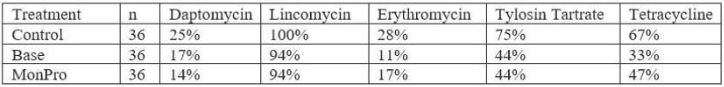
Table 1. Percentage of resistance for specific antibiotics across treatment.
While the enterococci isolated in this study, displayed resistance to at least one antibiotic in all three treatments, there were differences in the MDR. The most MDR was observed in the control diet which was supplemented with tylosin. Isolates with the MDR collected from the cattle fed the base diet was similar to the isolates from cattle fed the MonPro diet. Supplementation with L28 instead of tylosin resulted in fewer MDR enterococci.
These data suggest the inclusion of L28 could potentially be an effective alternative to tylosin to reduce the presence of MDR bacteria in feedlot cattle.
The authors are affiliated with the International Center for Food Industry Excellence, Department of Animal and Food Sciences at Texas Tech University. NP
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!