2020 Food Safety Report | Science & Technology
The chemistry of peracetic acid
Bacterial pathogens, such as Salmonella spp. and Campylobacter, continue to be the leading suspected causes of foodborne illnesses from poultry products today. Currently, multiple intervention strategies in processing help reduce and eliminate these pathogens, with one fast becoming dominate over the others: peracetic acid (PAA). This article serves as an overview of the chemistry and interaction of PAA with materials common to poultry-processing plants.
This is by no means an exhaustive description of PAA, but serves as a brief introduction.

Table of Contents:
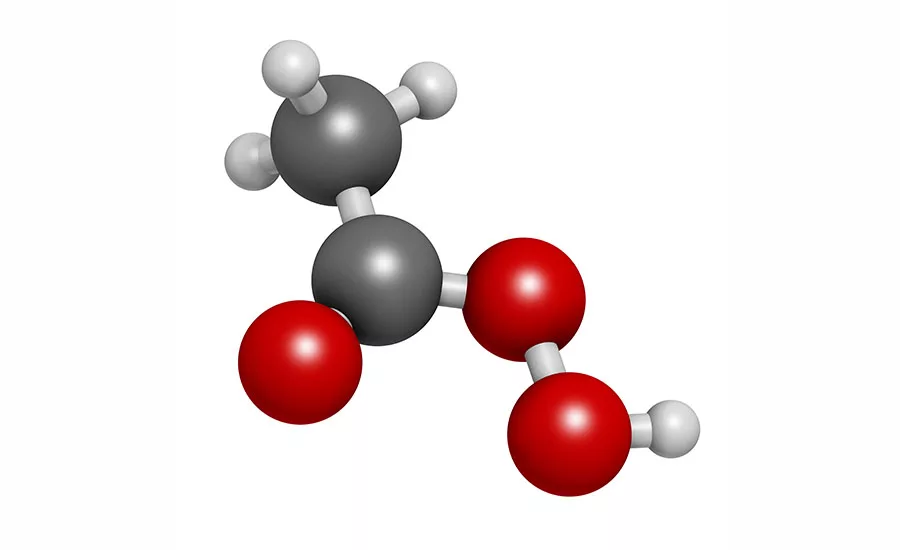
Peracetic acid, also known as peroxyacetic acid, is a peroxide version of acetic acid (vinegar) with an oxygen-oxygen bond. It is similar to hydrogen peroxide in structure, however, is more stable and a stronger antimicrobial compared to hydrogen peroxide due to the placement of this oxygen bond. For food applications, common formulations also usually contain a synthetic stabilizer to slow the rate of decomposition back into hydrogen peroxide and acetic acid. PAA is a colorless liquid with a characteristic pungent vinegar smell.
A PAA solution is a mixture of acetic acid, hydrogen peroxide, water and peracetic acid, which allows for many concentrations of PAA and hydrogen peroxide. Another reason for the various concentrations is its application. For example, the food industry typically uses a stock PAA with concentrations of 15-25 percent, while the paper industry requires a much higher concentration of PAA — around 50 percent — due to its bleaching ability.
While there are many modes of action for the disinfection activity of PAA, the main one is due to the release of the reactive oxygen species when PAA is activated by light in water. These reactive oxygen species cause irreparable damage to biomolecules common to bacteria as well as disruption to their normal cellular function.
The disinfection efficiency of PAA is dependent on certain factors that include temperature, pH, total suspended solids and metal ions.
PAA can function over a wide range of temperatures typical for the poultry industry; however, the rate of PAA degradation increases with temperature. It is sensitive to changes in pH and has its highest activity at or below 8.2. In general, PAA efficacy increases as the concentration of total suspended solids and biochemical oxygen demand decrease. The presence of metals, such as pure iron, will cause PAA to decay. Exposure to large amounts of metal in a short period of time can result in fire and/or explosions. Great care needs to be taken to avoid direct contact of PAA with iron metal.
Its efficacy for a wide range of pathogens makes PAA an attractive antimicrobial for the poultry industry. However, due to its reactive nature, great care is needed regarding how and where it will be stored and used within a facility. As the compatible and incompatible materials listed above are not exhaustive, managers should verify PAA material compatibility with equipment used throughout a plant by requesting a compatibility chart and consulting their PAA supplier. NP
Peracetic acid, at a glance
Advantages of PAA:
- Antimicrobial agent for a wide range of bacteria, viruses, spores, fungi and protozoan cysts
- Effective antimicrobial agent over a wide range of temperatures and pHs up to 8.5
- Decomposes into innocuous products of acetic acid, water and oxygen
- Compatible with some commonly used materials such as stainless steel, polytetrafluoroethylene (PTFE), and polyvinylidene fluoride (PVDF)
Disadvantages of PAA:
- Strict storage conditions to prevent fire
- Cost
- Corrosive to copper, copper alloys, iron and synthetic rubber
- Decomposes into acetic acid, which increases chemical oxygen demand in wastewater
Looking for a reprint of this article?
From high-res PDFs to custom plaques, order your copy today!



